This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
What are Protein Domains?
Protein domains are distinct units within a protein. They have a distinct structure when compared to the other subunits of the protein. This is because structure is imperative for protein function. All of the protein domains perform a unique function that, combined with the other domains add up to the full function of the protein. [1]
Understanding protein domains is an extremely important part of understanding overall protein function. Domains can provide information about the function of a protein, what other molecules it interacts with, and what proteins in other organisms may be homologous with it. [2]
Understanding protein domains is an extremely important part of understanding overall protein function. Domains can provide information about the function of a protein, what other molecules it interacts with, and what proteins in other organisms may be homologous with it. [2]
MTHFR Domains
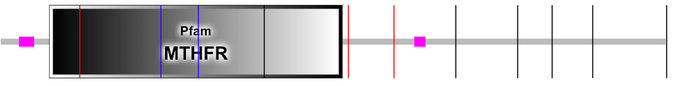
MTHFR domains in humans were identified using the data analysis tools Pfam, SMART, and InterPro. It is common to have discordance in the literature about protein domains, so it is imperative to use multiple tools to check for agreement.
Concordance was reached, and the Pfam and SMART domain analysis results are pictured. InterPro did not have the MTHFR domain characterized. MTHFR has one domain which is also named MTHFR.
Concordance was reached, and the Pfam and SMART domain analysis results are pictured. InterPro did not have the MTHFR domain characterized. MTHFR has one domain which is also named MTHFR.
What is the Structure and Function of the MTHFR Domain?
|
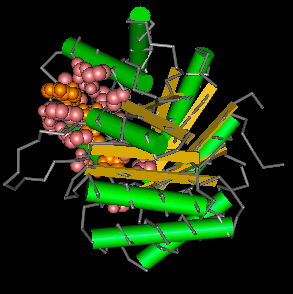
According to Pfam, MTHFR has a TIM barrel structure. This means that 8 alpha helices and eight parellel beta sheets are aligned to create a circular opening through the middle of the protein. This results in a barrel shape.
The MTHFR domain functions as a reducer. This means that it transfers electrons to another molecule or protein to slightly change that molecules structure and chemical properties. The MTHFR protein is documented in the literature to reduce 5,10-methylenetetrahydrofolate. [3] |
What Does the MTHFR Domain Look Like in Human Homologs?
Pictured above are the domains of MTHFR homologs in important model organisms as identified by Pfam. The MTHFR domain itself is very well-conserved in all of the homologs investigated. The only protein that looks significantly different from the others is Drosophila melanogaster which has a significant truncation.
Discussion
The presence of the single MTHFR domain on MTHFR suggests that most of its function can be localized to this region of the protein. Additionally, its high conservation across homologs suggests that it has been advantageous for survival and positively selected for throughout evolution. This domain is also likely an important contributor to disease pathogenesis. This notion is supported by research which indicates that all pathogenic mutations contributing to spina bifida identified so far are located within this region. [4]
References:
[1] European Bioinformatics Institute. (n.d.) What are Protein Domains? Retrieved from: https://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
[2] NCBI. (2013, February 12). Using Conserved Domains to Find Protein Homologs. NCBI Insights. Retrieved from: https://ncbiinsights.ncbi.nlm.nih.gov/2013/02/12/using-conserved-domains-to-find-functional-homologs/
[3] NCBI. (2019, March 12). MTHFR methylenetetrahydrofolate reductase [Homo sapiens (human)].
[4] Shaw, G.M., Lu, W., Zhu, H., Yang, W., Briggs, F.B., Carmichael, S.L., Barcellos, L.F., Lammer, E.J. & Finnell, R.H. (2009). 118 SNPs of folate- related genes and risks of spina bifida and conotruncal heart defects. Biomedical Central Medical Genetics, 10, 49. doi: 10.1186/1471-2350-10-49
Images:
Header: https://www.kuhnlab.bmb.msu.edu/
Figure 1: http://pfam.xfam.org/family/PF02219.17
Figure 2: http://smart.embl-heidelberg.de/smart/job_status.pl?jobid=1732820825116471552619764MfEzAROBgL
Image: https://www.ncbi.nlm.nih.gov/Structure/cdd/cd00537
[1] European Bioinformatics Institute. (n.d.) What are Protein Domains? Retrieved from: https://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
[2] NCBI. (2013, February 12). Using Conserved Domains to Find Protein Homologs. NCBI Insights. Retrieved from: https://ncbiinsights.ncbi.nlm.nih.gov/2013/02/12/using-conserved-domains-to-find-functional-homologs/
[3] NCBI. (2019, March 12). MTHFR methylenetetrahydrofolate reductase [Homo sapiens (human)].
[4] Shaw, G.M., Lu, W., Zhu, H., Yang, W., Briggs, F.B., Carmichael, S.L., Barcellos, L.F., Lammer, E.J. & Finnell, R.H. (2009). 118 SNPs of folate- related genes and risks of spina bifida and conotruncal heart defects. Biomedical Central Medical Genetics, 10, 49. doi: 10.1186/1471-2350-10-49
Images:
Header: https://www.kuhnlab.bmb.msu.edu/
Figure 1: http://pfam.xfam.org/family/PF02219.17
Figure 2: http://smart.embl-heidelberg.de/smart/job_status.pl?jobid=1732820825116471552619764MfEzAROBgL
Image: https://www.ncbi.nlm.nih.gov/Structure/cdd/cd00537